You encounter 4S2P in battery terminology when specifying lithium battery packs for medical devices. This configuration shapes voltage and capacity, which directly impacts safety and device performance. Selecting the right chemistry, voltage, and capacity ensures reliable operation in critical medical environments.
Chemistry | Platform Voltage (V) | Energy Density (Wh/kg) | Cycle Life (cycles) | Typical Applications |
|---|---|---|---|---|
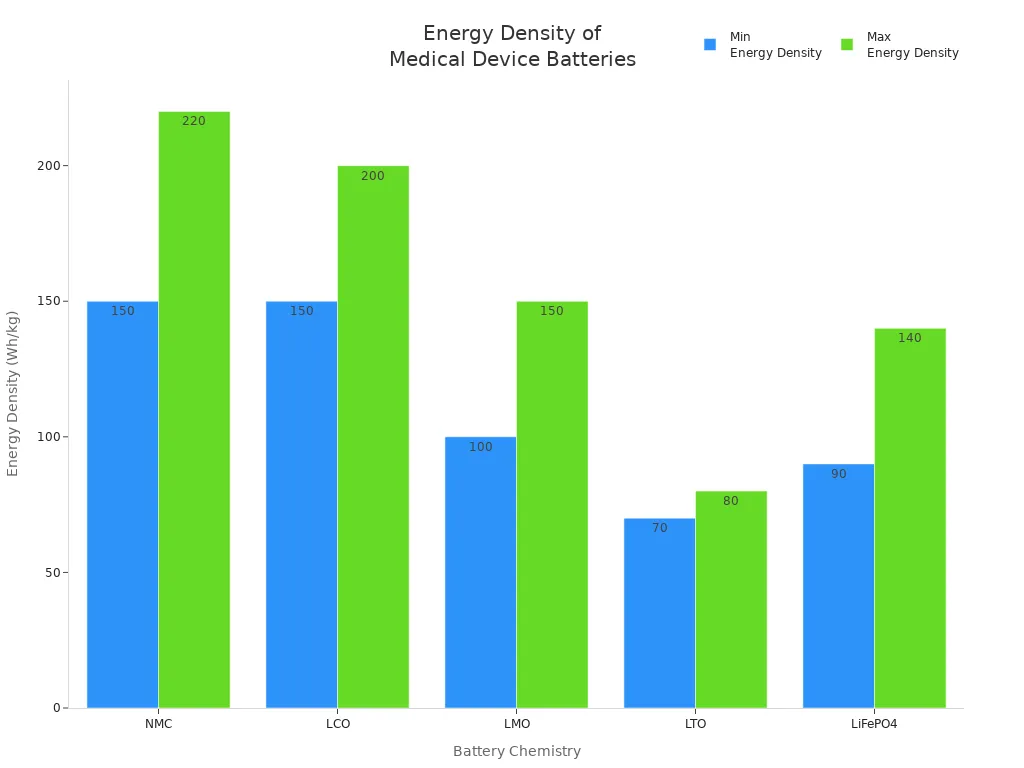
NMC | 3.7 | 150–220 | 1,000–2,000 | Medical, robotics, security |
LCO | 3.7 | 150–200 | 500–1,000 | Consumer electronics, medical |
LMO | 3.7 | 100–150 | 300–700 | Medical, industrial, power tools |
LTO | 2.4 | 70–80 | 5,000–10,000 | Industrial, infrastructure, medical |
LiFePO4 | 3.2 | 90–140 | 2,000–5,000 | Medical, robotics, security |
You must follow regulatory standards and integrate protection circuit modules (PCMs) to maximize medical device safety and reliability.
Key Takeaways
Understanding 4S2P configurations helps ensure reliable power for medical devices. This setup combines four cells in series and two in parallel, balancing voltage and capacity for optimal performance.
Regularly monitor the state-of-health and state-of-charge of medical batteries. This practice prevents unexpected failures and enhances patient safety by allowing for proactive maintenance.
Comply with safety standards and integrate protection circuit modules (PCMs) in battery packs. These measures ensure safety, reliability, and compliance with regulations in medical environments.
Part1: Battery Terminology and 4S2P Basics

1.1 What is 4S2P in Battery Terminology
You encounter the term 4S2P frequently when specifying battery packs for medical devices. In battery terminology, 4S2P means you have four cells connected in series and two cells connected in parallel. This configuration uses a total of eight cells. When you use standard 18650 lithium ion batteries, each cell has a nominal voltage of 3.7V. Connecting four cells in series gives you a combined voltage of 14.8V. The parallel connection doubles the capacity, which is critical for medical devices that require extended runtime and stable performance.
You select 4S2P configurations for mid-power medical applications because they balance voltage and capacity. This setup supports reliable operation in devices such as portable monitors, infusion pumps, and diagnostic equipment. You benefit from increased battery performance and reduced costs for battery management systems.
Tip: You should always verify the total number of cells and the configuration when choosing battery packs for medical devices. This ensures you meet both voltage and capacity requirements for your application.
Advantages of 4S2P in Medical Devices:
You achieve higher reliability in battery packs.
You reduce costs associated with battery management systems.
You combine series and parallel connections to enhance output voltage and increase capacity.
1.2 Series and Parallel Connections Explained
You need to understand how series and parallel connections affect battery performance in medical devices. When you connect cells in series, you add their voltages together. For example, four lithium ion batteries in series provide 14.8V. The capacity remains the same as a single cell. In parallel, you keep the voltage the same but double the capacity. Two cells in parallel provide twice the capacity, which means longer runtime for your medical devices.
Table: Impact of Series and Parallel Connections on Battery Performance
Configuration Type | Impact on Voltage | Impact on Capacity | Overall Performance |
|---|---|---|---|
Series | Voltage increases with each cell added | Capacity remains the same | Terminal voltage decreases with aging cells; low energy utilization if cells differ |
Parallel | Voltage remains the same | Capacity increases with each cell added | Runtime increases; weak cells reduce total load capability; risk of overdischarge |
You must monitor battery packs closely. In parallel circuits, a failing cell reduces load capability but does not affect voltage. In series, a failing cell can cause significant performance issues, similar to an engine running on fewer cylinders. High resistance cells are less critical in parallel, but a shorted cell can cause excessive heat and fire hazards. You should always use protection circuit modules to manage these risks in medical devices.
Note: Monitoring voltage and current limits in series and parallel configurations is essential for maintaining battery performance and safety in medical applications.
1.3 Key Specifications: Chemistry, Voltage, Capacity
You must evaluate several key specifications when selecting battery packs for medical devices. Chemistry, voltage, and capacity determine battery performance, safety, and reliability. You often choose lithium ion batteries for medical devices because they offer long service life and high energy density. You may also consider LiFePO4, lithium-polymer/LiPo, or solid-state battery chemistries for specialized applications.
Table: Comparison of Battery Chemistries for Medical Devices
Battery Chemistry | Advantages | Limitations |
|---|---|---|
Lithium-ion | Long service life, rechargeable | N/A |
LiFePO4 | High safety, long cycle life | Lower energy density |
Lithium-polymer/LiPo | Flexible form factor, lightweight | Sensitive to overcharge |
Solid-State Battery | Enhanced safety, high energy density | Emerging technology, higher cost |
Nickel-Metal Hydride | Rechargeable, drop-in replacement for alkaline | Lower energy density |
Lead-acid | Low cost, reliable for stationary use | Shorter lifespan, potential leakage |
You must match battery voltage and capacity to the requirements of your medical devices. Nominal capacity, internal resistance, self-discharge rate, cycle life, and temperature coefficient all influence battery performance and device reliability.
Table: Battery Specifications for Portable Medical Equipment
Specification | Importance |
|---|---|
Nominal Capacity (mAh) | Baseline energy; consider discharge profile |
Internal Resistance (mΩ) | Affects heat generation and cell balancing |
Self-Discharge Rate | Ensures device readiness after storage |
Cycle Life at Partial DoD | Predicts longevity under typical device use |
Temperature Coefficient | Maintains performance in medical-grade operating environments |
You must optimize these parameters for each medical device. Device runtime and operational capabilities depend on voltage, capacity, and self-discharge rates. You should always review supplier specification sheets and conduct customized testing to ensure batteries meet operational, reliability, and safety requirements. Compliance with industry standards and governmental regulations increases costs but ensures safe and effective medical devices.
Alert: You face challenges such as power loss, overheating, and chemical exposure when designing battery packs for medical devices. Sudden power loss can interrupt patient monitoring. Overheating can cause fires or explosions. Leaking batteries expose patients and healthcare workers to harmful substances. You must address these risks through careful selection and management of battery packs.
You must understand the types of batteries available and select the best chemistry, voltage, and capacity for your medical devices. This ensures optimal battery performance, safety, and reliability in critical healthcare environments.
Part2: Medical Battery Health, Safety, and Compliance

2.1 State-of-Health and State-of-Charge Monitoring
You must monitor the state-of-health and state-of-charge of every medical battery to ensure reliable operation in medical devices. The state-of-health describes the current condition of a battery compared to its original specifications. The state-of-charge indicates how much charge remains relative to full capacity. You need to track both absolute state-of-health and absolute state-of-charge to prevent unexpected failures in critical medical environments.
You can use several methods to monitor these parameters. Each method offers unique advantages and limitations for medical battery management.
Method | Description | Pros | Cons |
|---|---|---|---|
Coulomb Counting | Tracks the flow of current in and out of the battery. | Accurate when the initial state-of-charge is known; useful for monitoring over time. | Errors accumulate without recalibration; highly sensitive to measurement noise. |
Voltage-Based Method | Estimates state-of-charge by comparing the battery’s voltage against reference values. | Simple, cost-effective, and quick. | Affected by load conditions, temperature changes, and battery chemistry—making it less accurate. |
Impedance Spectroscopy | Sends a small AC signal through the battery and measures resistance to estimate state-of-charge. | Provides detailed insights into the internal battery state. | Requires specialized equipment and is not practical for everyday applications. |
Capacity Testing | Fully charge and discharge the battery to measure actual capacity versus original specifications. | Direct measurement of health. | Time-consuming and may not reflect real-world usage. |
Internal Resistance Measurement | Measures resistance to assess battery health. | Simple and effective indicator of health. | Requires accurate current and voltage measurements. |
Self-Discharge Rate | Evaluates how quickly a battery loses charge when idle. | Simple to measure. | High self-discharge indicates poor health and potential internal damage. |
You should implement real-time battery monitoring in all medical devices. This approach gives you continuous visibility into battery health and state-of-charge. You can detect issues early, schedule proactive maintenance, and receive instant alerts if performance data shows degradation. These practices help you avoid device failure and improve patient safety.
Tip: Always use advanced battery management systems to track both relative state-of-health and relative state-of-charge. This ensures you maintain optimal medical battery performance and extend device lifespan.
2.2 Performance and Reliability in Medical Devices
You depend on battery performance and reliability to deliver consistent results in medical devices. Battery failure remains the leading cause of malfunctions in many medical applications, including implantable defibrillators. In one study, battery failures accounted for 1.42% of all device malfunctions. In another analysis, 57.9% of malfunctions in implantable defibrillators resulted from battery issues. Rapid battery depletion due to internal short-circuiting has led to patient harm and even fatalities.
You must monitor the state-of-health and state-of-charge to prevent these failures. Real-time monitoring allows you to identify weak batteries before they fail. You can schedule preventive maintenance and replace batteries proactively. Remote monitoring systems provide instant alerts when battery health declines, allowing you to intervene before device performance suffers.
You should also consider how user practices and device usage patterns affect battery lifespan. The following table summarizes key factors:
Factor | Impact on Battery Lifespan |
|---|---|
Frequency of Use | Continuous operation depletes batteries faster than intermittent use. |
Depth of Discharge | Deep discharges reduce lifespan significantly; below 20% can lead to 30-40% fewer cycles. |
Charging Practices | Overcharging and irregular cycles accelerate degradation. Using original chargers is recommended. |
Environmental Conditions | High temperatures cause capacity loss; humidity can lead to corrosion. |
Storage Practices | Storing fully discharged batteries can cause irreversible damage; recommended storage is at 40-60% charge in cool, dry conditions. |
You must optimize these factors to maximize medical battery reliability and ensure uninterrupted operation of medical devices.
2.3 Safety Standards and Regulatory Compliance
You must comply with international safety standards to ensure medical battery safety in all medical devices. The following table outlines the most relevant standards and their key requirements:
Standard | Key Compliance Requirements |
|---|---|
IEC 60601-1 | General requirements for the basic safety and essential performance of medical electrical equipment, including batteries. |
IEC 62133 | Safety of secondary cells and batteries, including biocompatibility and safety features for proximity to patients. |
UL 2054 | Safety standard for batteries covering electrical, mechanical, environmental, and thermal safety. |
ISO 13485 | Quality management system requirements for safe and reliable battery production. |
ISO 10993-1 | Guidelines for evaluating biological safety, assessing risks related to cytotoxicity and genotoxicity. |
Regulatory agencies enforce these standards through strict requirements. You must ensure that every medical battery meets the following criteria:
Requirement | Description |
|---|---|
FDA General Safety and Performance Requirements | Batteries must meet the safety rules of IEC 62133, UL 2054, ISO 13485, and IEC 60601-1. |
Biocompatibility | Batteries must be safe to use in the body. |
Safety Features | Batteries need special safety parts for use near people. |
Authentication | Batteries must be checked to stop fake ones. |
Serialization | Batteries must have numbers so you can track them. |
Transportation | Medical batteries must follow all shipping rules. |
Design Features | Batteries need things like overcharge protection, thermal shutdown, and biocompatibility. |
You must also test batteries for altitude simulation, thermal stability, vibration, shock, external short circuit, impact/crush, overcharge, and forced discharge. These tests ensure that lithium battery packs meet all safety standards for medical devices.
You face additional challenges when manufacturing for global markets. Different countries have unique definitions and classifications for medical devices, which affect battery safety regulations. You must adapt your compliance strategies to meet the specific requirements of each region. This can increase costs and complicate global distribution, but it is essential for market access and patient safety.
2.4 Role of PCMs and Risk Management
You must integrate protection circuit modules (PCMs) into every medical battery pack to manage risk and ensure safety. PCMs provide multiple layers of protection for lithium battery packs in medical devices. Their functions include:
Function | Description |
|---|---|
Overcharge Protection | Prevents charging beyond maximum voltage limit to avoid overheating and potential explosions. |
Over-Discharge Protection | Prevents discharging below minimum voltage threshold to protect battery chemistry and lifespan. |
Overcurrent and Short Circuit | Protects against excessive current draw during discharge or charging, using components like MOSFETs. |
Temperature Protection | Monitors temperature to prevent operation if the battery becomes too hot. |
Cell Balancing | Ensures even charging and discharging in multi-cell packs for optimal performance. |
Voltage and Current Monitoring | Continuously monitors voltage and current to detect unsafe conditions. |
Automatic Disconnect | Disconnects battery from load or charger when unsafe parameters are detected. |
Reconnection | Allows reconnection once parameters return to safe levels. |
LED Indicators | Provides visual status indicators for the battery’s condition. |
Communication with Devices | Advanced systems can communicate battery status to the powered device. |
You can learn more about PCMs and battery management systems at BMS and PCM.
You should also implement robust risk management strategies for medical battery safety. These include failure analysis, regulatory compliance, manufacturing quality control, and postmarket surveillance. Failure analysis helps you understand the root causes of battery failures and prevent recurrence. Regulatory compliance ensures you meet all safety standards. Quality control during manufacturing reduces defects. Postmarket surveillance allows you to monitor battery performance and address emerging risks.
2.5 Maintenance and Disposal of Medical Batteries
You must follow best practices for maintenance and disposal to extend the lifespan of every medical battery and protect the environment. For maintenance, you should:
Maintain stable charging habits by keeping batteries between 20% and 80% charge.
Reduce exposure to heat by storing batteries at 25°C (77°F) or below with proper ventilation.
Rotate batteries in high-demand devices to distribute wear evenly.
Schedule preventive testing every 6–12 months to monitor battery health.
Inspect batteries for physical wear, such as swelling or corrosion.
Store backup batteries at 40–60% charge in a cool, dry location.
Use OEM-compliant chargers to avoid voltage irregularities.
Train staff on proper handling procedures to recognize early warning signs.
For disposal, you must comply with all environmental and safety regulations. In the United States, the EPA classifies most spent lithium-ion batteries as hazardous waste under the Resource Conservation and Recovery Act (RCRA). The Department of Transportation regulates the transport of hazardous materials, including lithium batteries. In the European Union, manufacturers must design products for recycling under extended producer responsibility rules starting in 2030. You should never crush lithium-ion batteries or mix different chemistries, as this can cause fires. Safe disposal practices prevent risks to patients, staff, and the environment.
Alert: Improper maintenance or disposal of medical batteries can lead to device failure, environmental harm, and regulatory penalties. Always follow industry guidelines and train your team on best practices.
By prioritizing state-of-health and state-of-charge monitoring, adhering to safety standards, integrating PCMs, and following proper maintenance and disposal protocols, you ensure the highest level of medical battery safety and reliability in all medical devices.
You improve health outcomes by understanding battery terminology and 4S2P configuration for medical devices. This knowledge ensures lithium battery packs deliver consistent power, long shelf life, and compact design.
Feature | Description |
|---|---|
Consistent Power | Maintains voltage within ±0.1V under load, crucial for accurate readings. |
Long Shelf Life | Holds charge for months without degradation, ideal for emergency reserves. |
Compact Design | Enables integration into space-constrained medical devices. |
You benefit from proper battery management and compliance with safety standards:
Improved patient health through reliable device performance.
Reduced risk of device failure in medical environments.
Compliance with regulatory standards for market access.
Enhanced reliability of lithium battery-powered medical devices.
You should collaborate with battery experts and follow regulatory guidelines to ensure the safety and performance of lithium battery packs in all medical devices.
FAQ
What does 4S2P mean for lithium battery packs in medical devices?
You use 4S2P to describe four lithium cells in series and two in parallel. This configuration increases voltage and capacity for reliable medical device operation.
How do you select the best lithium battery chemistry for medical, robotics, or security systems?
You compare lithium chemistries using cycle life, energy density, and safety. For example, NMC offers high energy density, while LiFePO4 provides longer cycle life and enhanced safety.
Chemistry | Energy Density (Wh/kg) | Cycle Life (cycles) | Safety Level |
|---|---|---|---|
NMC | 150–220 | 1,000–2,000 | High |
LiFePO4 | 90–140 | 2,000–5,000 | Very High |
Where can you find custom lithium battery solutions for industrial or medical applications?
You can contact Large Power for custom lithium battery packs.




