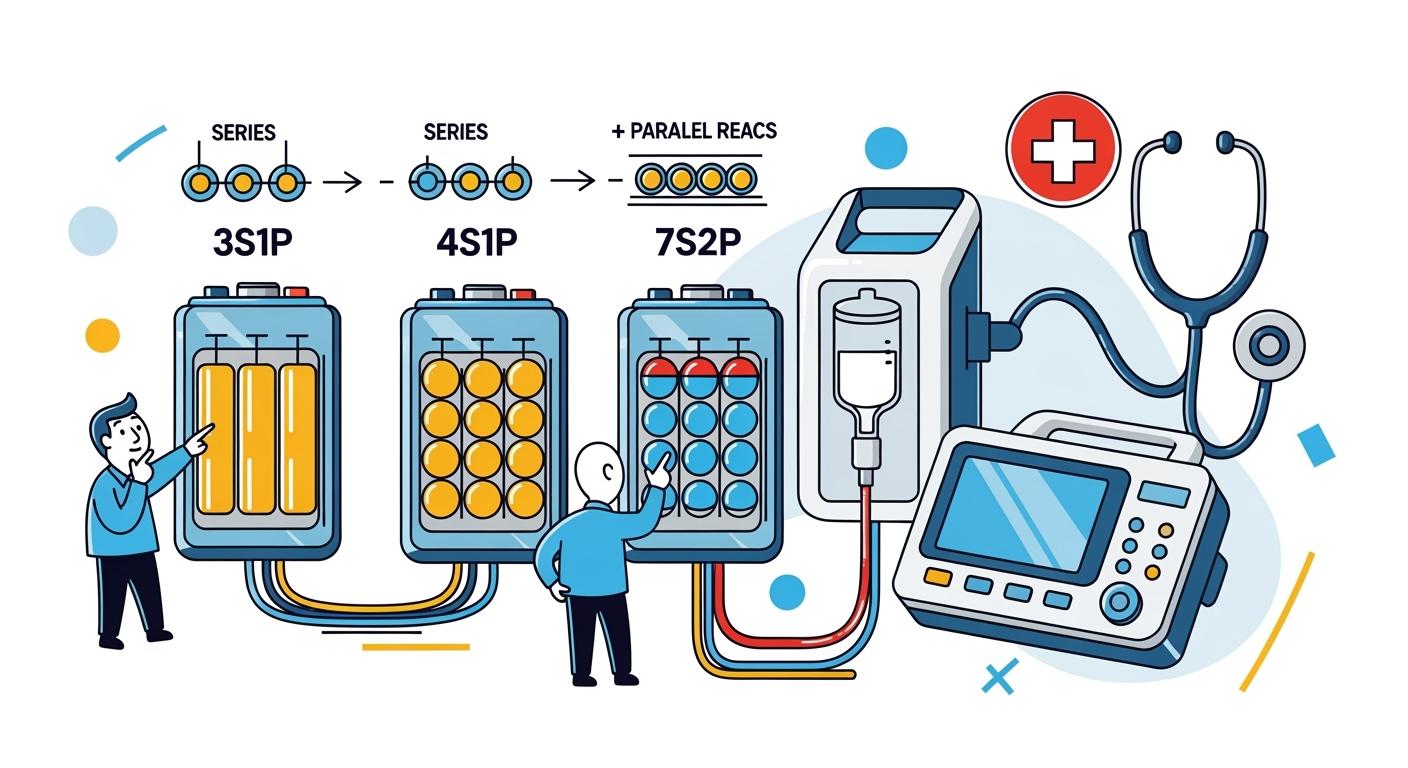
You choose 3s1p, 4s1p, or 7s2p battery packs by matching the configuration to your devices’ energy needs, voltage, and safety requirements. Medical devices demand reliable lithium batteries for medical use, predictable performance, and longer service life. Regulatory compliance and quality design ensure reliability for diagnostic and imaging equipment, implantable applications, and portable medical devices. Battery management systems and custom lithium-ion batteries support diagnostics, custom lithium battery solutions, and high-quality power systems.
Regulatory Compliance Table: IEC 60601 Impact on Battery Selection
Requirement | Impact on Medical Battery Packs |
|---|---|
Safety & Performance | Minimizes electrical/mechanical hazard risks |
Market Access | Certification delays/block market entry if unmet |
Risk Management | Requires risk identification, quantification, mitigation |
Key Takeaways
Choose the right battery configuration (3S1P, 4S1P, or 7S2P) based on your medical device’s voltage and capacity needs to ensure optimal performance.
Consider regulatory compliance and safety standards, such as IEC 60601, to avoid certification delays and ensure patient safety.
Evaluate runtime and size constraints when selecting battery packs, as portable devices require compact batteries with sufficient power for extended use.
Part1: Battery Pack Basics for Medical Devices
1.1 3S1P, 4S1P, 7S2P Explained
You encounter several battery pack configurations when selecting batteries for devices in the medical field. Each configuration impacts the performance and suitability of medical device batteries. The 3S1P, 4S1P, and 7S2P arrangements refer to how cells are connected within lithium battery packs. In a 3S1P pack, three cells connect in series, providing a higher voltage for devices that require more power. The 4S1P configuration uses four cells in series, offering even greater voltage for advanced medical devices. The 7S2P pack combines seven cells in series and two in parallel, delivering both high voltage and increased capacity for demanding medical applications.
You should understand how these configurations affect your devices. The 7S2P pack, for example, achieves a total voltage of 168 volts and a capacity of 84.8 ampere-hours, making it suitable for high-power medical devices.
Configuration | Series (S) | Parallel (P) | Total Voltage | Total Capacity |
|---|---|---|---|---|
3S1P | 3 | 1 | 3 cells voltage | 1 cell capacity |
4S1P | 4 | 1 | 4 cells voltage | 1 cell capacity |
7S2P | 7 | 2 | 7 cells voltage | 2 cells capacity |
You often see lithium-ion, LiFePO4, lithium-polymer/LiPo, and solid-state battery chemistries used in these packs. Each chemistry offers unique benefits for medical devices.
1.2 Series vs. Parallel in Medical Device Batteries
You must know how series and parallel arrangements influence medical device batteries. Connecting cells in series increases the total voltage, which is essential for devices that need higher operating voltages. The capacity remains the same as a single cell. When you connect cells in parallel, the voltage stays constant, but the capacity increases, allowing devices to run longer between charges.
Series configuration: Raises voltage for devices needing more power.
Parallel configuration: Boosts capacity for devices requiring extended operation.
Combining both: Lets you tailor medical device batteries to meet specific voltage and capacity needs.
You rely on these configurations to optimize battery performance for medical devices. Understanding these basics helps you select the right lithium battery packs for your devices and ensures reliable operation in critical medical environments.
Part2: Compare and Choose 3S1P, 4S1P, 7S2P for Devices

2.1 Voltage, Capacity, Size Overview
When you choose 3s1p, 4s1p, or 7s2p battery packs for medical devices, you must evaluate voltage, capacity, and size. These factors determine how well medical device batteries support your devices in clinical environments. You need to match the battery configuration to the device’s operating voltage and runtime requirements. The following table summarizes the typical voltage and capacity ranges for medical device batteries using lithium-ion and lithium-polymer chemistries:
Parameter | 3S1P Battery Pack | 4S1P Battery Pack | 7S2P Battery Pack |
|---|---|---|---|
Nominal Voltage | 11.1V (Li-ion/LiPo) | 14.8V (Li-ion/LiPo) | 25.9V (Li-ion/LiPo) |
Typical Capacity | 2.2Ah – 3.4Ah | 2.5Ah – 3.5Ah | 4.4Ah – 7.0Ah |
Physical Size | Compact | Moderate | Larger, modular |
You see that 3s1p battery packs offer lower voltage and capacity, making them suitable for portable devices with limited space. The 4s1p configuration provides a moderate increase in both voltage and capacity, supporting devices that require more power but still need a compact form factor. The 7s2p battery pack delivers the highest voltage and capacity, ideal for medical devices that demand extended operation and higher energy output.
2.2 Typical Medical Device Applications
You must choose 3s1p, 4s1p, or 7s2p battery packs based on the specific needs of your devices. Medical device batteries play a critical role in ensuring reliable performance and patient safety. Here are examples of how each configuration fits different medical applications:
3s1p battery packs power portable infusion pumps, handheld diagnostic scanners, and wearable monitors. These devices require lightweight batteries and moderate energy output.
4s1p battery packs support advanced patient monitoring systems, portable ventilators, and imaging accessories. These devices need higher voltage and longer runtime than basic portable devices.
7s2p battery packs serve surgical robots, mobile X-ray machines, and high-capacity backup systems. These devices demand robust medical device batteries with maximum voltage and capacity for continuous operation.
You must assess the energy requirements and physical constraints of your devices before you choose 3s1p, 4s1p, or 7s2p battery packs. Medical device batteries must deliver consistent performance and meet regulatory standards for safety and reliability.
Tip: Custom battery designs can extend service life and reduce replacement frequency. You benefit from predictable performance and easier maintenance, which lowers the total cost of ownership for your medical devices.
Longer service life and fewer replacements mean you spend less on battery turnover.
Predictable performance ensures your devices operate reliably in real-world settings.
Modular battery systems allow quick replacements, reducing downtime and maintenance costs.
2.3 Key Differences Summary
You need to understand the key differences between 3s1p, 4s1p, and 7s2p battery packs to make informed decisions for your medical devices. Medical device batteries differ in voltage, capacity, size, and suitability for various applications.
Feature | 3S1P Battery Pack | 4S1P Battery Pack | 7S2P Battery Pack |
|---|---|---|---|
Voltage | Low | Moderate | High |
Capacity | Low | Moderate | High |
Size | Small | Medium | Large |
Application | Portable, wearable | Monitoring, ventilators | Surgical, imaging, backup |
Cost of Ownership | Lower | Moderate | Higher (but longer life) |
You choose 3s1p battery packs for compact, low-power devices. You select 4s1p battery packs for devices that need more energy and longer runtime. You rely on 7s2p battery packs for high-power medical devices that require maximum capacity and voltage.
You must align battery configuration with your device’s operational needs, safety standards, and maintenance goals. Medical device batteries that match your requirements will improve reliability, reduce costs, and enhance patient care.
Part3: Selection Factors for Medical Device Batteries

3.1 Device Voltage and Power Needs
You must start your selection process by analyzing the voltage and power needs of your devices. Each medical device has unique requirements for voltage, current, and power output. You should reference the IEC 60601 standard, which defines power supply requirements and risk assessment for medical devices. This standard also addresses insulation, electromagnetic compatibility, and leakage current, all of which impact battery selection.
Method/Aspect | Description |
|---|---|
IEC 60601 Standard | Focuses on safety, risk assessment, and power supply requirements for medical devices. |
Risk Assessment Types | Classifies devices as body (B), body floating (BF), or cardiac floating (CF). |
Means of Protection (MOP) | Defines safety measures for patient (MOPP) and operator (MOOP). |
Insulation Classifications | Includes functional, protective, basic, supplementary, double, and reinforced insulation. |
Electromagnetic Compatibility | Addresses risks from electromagnetic interference. |
Minimum Isolation Voltage | Specifies the minimum isolation voltage for safety compliance. |
Leakage Current Considerations | Evaluates risks related to patient and operator exposure. |
You must calculate the total power consumption of your devices. Devices with lower power needs, such as portable monitors, often use 3S1P battery packs. Devices with higher power requirements, like advanced diagnostics or imaging systems, may require 4S1P or 7S2P configurations. You should always match the battery pack voltage to the device’s operating voltage to ensure optimal performance and regulatory compliance.
3-cell batteries work best for portable devices with low power demands.
4-cell batteries support higher power consumption and longer usage times.
7S2P configurations balance high voltage and capacity for demanding applications.
You must consider the regulatory readiness of your devices. This includes ensuring that your battery packs meet all applicable standards for voltage, insulation, and electromagnetic compatibility.
3.2 Runtime, Capacity, and Size
You must evaluate runtime, capacity, and size when selecting medical device batteries. Devices used in mobile or home healthcare settings require long runtimes and compact battery packs. You must balance extended runtime with size constraints, as larger batteries can reduce portability and usability.
The demand for portable medical devices has increased due to patient mobility needs.
Reliability and safety take priority over energy density and form factor.
You face challenges in balancing runtime with size, which impacts device portability.
Thermal management is critical, as temperature fluctuations affect battery performance and safety.
You should select battery packs that provide sufficient runtime for your devices without compromising portability. For example, 3S1P battery packs offer compact size and moderate capacity, making them ideal for handheld devices. 4S1P and 7S2P packs deliver higher capacity and longer life, supporting devices that require extended operation.
Configuration | Typical Runtime | Size | Application Example |
|---|---|---|---|
3S1P | Short | Compact | Portable infusion pumps |
4S1P | Moderate | Moderate | Patient monitoring systems |
7S2P | Long | Larger | Mobile X-ray, surgical robots |
You should also consider modularity in battery design. Modular battery packs allow for easy replacement and maintenance, reducing downtime and operational costs. Standardization of modules enhances flexibility and extends the life of your devices.
3.3 Safety, BMS, and Regulatory Compliance
You must prioritize safety, BMS integration, and regulatory compliance when selecting medical device batteries. Safety features are essential for protecting both patients and operators. You should ensure that your battery packs undergo rigorous testing, including high-altitude simulation, thermal cycling, vibration, mechanical shock, and crush/impact testing.
Safety Feature | Standard/Requirement |
|---|---|
High-altitude simulation | Required for performance under pressure |
Thermal cycling | Tests battery performance across temperatures |
Vibration testing | Ensures mechanical robustness |
Mechanical shock | Assesses durability against impacts |
Crush/impact testing | Evaluates structural integrity |
IEC 62133 | International lithium-ion battery safety |
UL 2054 | Fire exposure and enclosure integrity |
FDA and ISO Compliance | Documentation and testing |
ISO 13485 and ISO 9001 | Quality and reliability management |
Batch validation | Prevents failures and ensures safety |
You must integrate a battery management system (BMS) into your custom lithium battery solutions. A BMS prevents overheating, overcharging, and deep discharge, extending battery life and improving reliability. It also provides real-time monitoring, allowing you to predict remaining runtime and avoid unexpected shutdowns. BMS integration supports regulatory compliance by facilitating certifications and traceability.
BMS enhances safety by preventing fires and failures.
It extends battery life by managing charge and temperature.
It improves device reliability and supports regulatory documentation.
You must ensure compliance with all relevant standards, such as IEC 62133, UL 2054, ISO 13485, and IEC 60601-1. You should also address biocompatibility, serialization, and traceability to meet regulatory requirements. Compliance with regional standards, such as the EU Battery Regulation 2023/1542, is essential for market access.
Note: Regulatory compliance can be complex and time-consuming. You should work with experienced partners to streamline documentation, testing, and certification.
3.4 Battery Chemistry and Manufacturer Choice
You must select the appropriate battery chemistry for your devices. Lithium-ion batteries are widely used in medical device batteries due to their high energy density, long cycle life, and stable voltage output. You can also consider LiFePO4, lithium-polymer/LiPo, and solid-state battery chemistries for specific applications. For implantable devices, lithium thionyl chloride batteries offer long shelf life and exceptional reliability.
Chemistry Type | Energy Density (Wh/kg) | Cycle Life (cycles) | Self-Discharge Rate | Application Example |
|---|---|---|---|---|
Lithium-ion | 150–250 | Up to 1,000 | 2–3% per month | Portable monitors, diagnostics |
LiFePO4 | 90–120 | 2,000+ | <3% per month | Surgical tools, portable devices |
Lithium-polymer/LiPo | 100–200 | 500–1,000 | 3–5% per month | Wearables, compact devices |
Solid-State Battery | 250+ | 1,000+ | <1% per month | Next-gen implantable devices |
Lithium thionyl chloride | 400+ | 10+ years shelf | <1% per year | Implantable, long-life devices |
You can learn more about lithium-ion, LiFePO4, lithium-polymer/LiPo, and solid-state battery chemistries by visiting our resource pages.
You must also consider sustainability and conflict minerals when selecting battery packs. Manufacturers should comply with sustainability and conflict minerals standards to ensure ethical sourcing and environmental responsibility.
You should choose manufacturers with a proven track record in quality and reliability. Leading suppliers design custom lithium battery solutions with advanced safety features, proprietary technologies for sterilization, and compliance with ANSI/AAMI ES 60601-1 and IEC 62133. They also offer warranties and technical support, ensuring long-term reliability and life for your devices.
Manufacturers must integrate safety from cell selection to packaging.
Innovative design technologies protect batteries from extreme conditions.
Custom battery packs should meet all global certifications, including UL, CE, and UN38.3, to ensure market acceptance.
You select the right battery for medical devices by following these steps:
Calculate power needs and runtime for your devices.
Choose batteries with suitable chemistry and capacity.
Use chargers that match battery specifications.
Ongoing consultation with experts improves safety, compliance, and performance.
Emerging trends in medical batteries include smart management systems and wireless charging.
FAQ
What factors should you consider when selecting lithium battery packs for medical devices?
You must evaluate voltage, capacity, runtime, and safety. You should confirm regulatory compliance for medical applications. Large Power offers custom solutions for your devices.
How does a battery management system (BMS) improve safety in medical devices?
A BMS monitors temperature, voltage, and current. You prevent overheating and overcharging in your devices. This ensures safe operation for medical environments.
Can you request custom lithium battery packs for specialized medical devices?
You can request custom packs from Large Power. Visit custom battery consultation for tailored solutions that meet your medical device requirements.




