
You face significant risks when you neglect battery certification for your medical device battery. Regulatory standards help you guarantee the safety and reliability of lithium battery packs used in clinical environments. A structured certification approach protects your business from regulatory penalties and enhances trust with healthcare partners.
Key Takeaways
Battery certification is crucial for ensuring the safety and reliability of medical devices. It protects patients and enhances trust with healthcare partners.
Understanding regulatory standards like UL, CE, and IEC is essential. Each standard has unique requirements that impact market access and patient safety.
Robust documentation and traceability are vital for compliance. Maintain detailed records to support audits and ensure quick responses to safety issues.
Engage accredited laboratories for testing. Choose labs that meet international standards to ensure your battery meets safety and performance criteria.
Sustaining compliance is an ongoing process. Regular maintenance, documentation updates, and staff training are key to maintaining certifications and market access.
Part1: Battery Certification and Medical Device Safety
1.1 Lithium Battery Packs in Clinical Use
You rely on lithium battery packs to power a wide range of medical devices in clinical environments. These batteries support critical functions in portable ultrasound machines, compact imaging devices, and equipment that demands high safety standards. The table below compares the most common lithium battery chemistries used in medical applications:
Type of Battery | Advantages | Applications |
|---|---|---|
Lithium-ion Batteries | Exceptional energy density, long cycle life | Portable ultrasound, high-performance imaging |
Lithium Polymer Batteries | Lightweight, flexible form factor | Compact imaging devices |
Lithium Iron Phosphate Batteries | High safety, durability, resist overheating/fires | Devices requiring high safety |
Selecting the right battery type ensures your medical device battery meets operational demands and regulatory standards.
1.2 Safety and Reliability Requirements
You must meet strict battery requirements to achieve battery certification for your medical device battery. International regulatory bodies mandate safety certifications and quality management systems to protect patients and healthcare staff. The following table outlines key battery requirements and their impact on battery design:
Standard | Requirement | Impact on Battery Design |
|---|---|---|
IEC 62133 | Certification for rechargeable cells | Ensures safety and compliance for medical devices |
UL 2054 | Household/commercial battery certification | Requires rigorous safety testing and documentation |
ISO 13485 | Quality management system certification | Mandates thorough documentation and process control |
IEC 60601-1 | Safety testing for medical equipment | Requires batteries to meet performance criteria |
You must document every step and maintain process control to meet battery compliance certification and regulatory compliance.
1.3 Risks of Non-Compliance
Ignoring battery requirements exposes you to serious risks. Common hazards include short-circuiting, battery acid leaks, flammable gases, battery bursts, and physical hazards from weight. These incidents can occur in medical monitors, robotics, and portable equipment. You also face:
Product recall, leading to financial loss and reputation damage
Market ban, resulting in loss of global market access
Legal penalties, including fines and lawsuits
For example, ICU Medical recalled CSB batteries after discovering counterfeit units in critical devices. These batteries failed compatibility tests, causing device failures and interrupting essential medical treatments. You must prioritize lithium battery safety and battery compliance to protect patients and your business.
Part2: Key Regulatory Standards for Medical Device Batteries

2.1 UL, CE, and IEC Overview
You must understand the differences between UL standards, CE marking, and IEC certification when selecting battery certification for your medical device battery. Each standard addresses unique battery requirements and regulatory standards for global market access.
UL certification requires you to manufacture batteries in UL-certified facilities. You must follow strict limits on lithium content, which depend on whether the battery is technician-replaceable or user-replaceable. UL standards focus on safety certification and battery management systems to protect patient safety.
CE certification ensures your battery meets European regulatory standards for health, safety, and environmental protection. You must demonstrate compliance with battery safety standards and document all battery requirements.
IEC certification covers international regulatory standards for electrical and electronic devices. You must meet safety and performance criteria for battery pack certifications in medical applications.
Certification | Focus Area | Key Battery Requirements | Application |
|---|---|---|---|
UL Standards | Safety, facility certification, lithium content limits | Technician/user-replaceable battery requirements, battery management systems | North America, global manufacturers |
CE Marking | Health, safety, environmental protection | Compliance with EU regulatory standards, documentation | European Union, EEA |
IEC | International safety and performance | Battery pack certifications, battery management systems | Global, cross-border trade |
You must align your battery certification strategy with these regulatory standards to ensure patient safety and global market access.
2.2 ISO 13485 and Quality Management
ISO 13485 sets the quality management system requirements for manufacturing medical device batteries. You must implement strict controls, maintain robust documentation, and ensure full traceability throughout the production process. These regulatory standards help you prevent contamination and support continuous improvement.
Key Requirement | Description |
|---|---|
Stringent Controls | You must implement strict controls to ensure product safety and compliance with battery requirements. |
Robust Documentation | You need comprehensive documentation processes for tracking compliance and quality management system effectiveness. |
Cleanliness and Cross-Contamination | You must establish protocols to prevent contamination during battery manufacturing. |
Full Traceability | You must ensure products are traceable throughout the manufacturing process for accountability. |
Continuous Improvement | You must pursue ongoing enhancements in quality management system practices to maintain standards. |
Compliance with Regulatory Standards | You must adhere to regulatory standards for market acceptance and safety assurance. |
Industry studies show that compliance with ISO 13485 improves battery safety and reliability. You benefit from rigorous testing, quality control, and risk mitigation strategies that protect patient safety.
Evidence Description | Key Points |
|---|---|
Rigorous Testing and Quality Control | Compliance with ISO 13485 ensures thorough testing and quality control processes, enhancing battery safety and reliability. |
Regulatory Compliance | Adhering to ISO 13485 aligns with regulatory standards, which is crucial for medical device safety. |
Risk Mitigation Strategies | The standard helps in identifying and mitigating risks associated with battery failures, thus improving overall device safety. |
You must treat ISO 13485 as the foundation of your quality management system for battery certification.
2.3 IEC 62133 and Safety Testing
IEC 62133 is a critical international standard for battery certification. You must follow this standard to meet regulatory standards for rechargeable lithium-ion batteries in medical device batteries. IEC 62133 outlines battery requirements for electrical, mechanical, and chemical safety. You must conduct tests that address overcharging, over-discharging, short-circuiting, and thermal runaway to protect patient safety.
Test Type | Description |
|---|---|
Electrical Abuse Tests | You must evaluate the battery’s response to electrical faults such as overcharging and short-circuiting. |
Mechanical Abuse Tests | You must assess the battery’s durability against physical impacts and stress. |
Thermal Abuse Test | You must determine the battery’s behavior under extreme temperature conditions. |
You must integrate battery management systems that monitor and control these risks. IEC 62133 ensures your battery certification meets global regulatory standards and supports patient safety.
2.4 ANSI/AAMI ES 60601-1 and Essential Performance
ANSI/AAMI ES 60601-1 is a key standard for medical device battery safety in the United States and many global markets. You must ensure your battery certification meets the essential performance requirements outlined in this standard. The standard covers battery requirements for electrical safety, electromagnetic compatibility, and essential performance. You must design battery management systems that support patient safety and device reliability.
You must document all battery requirements and test your battery management systems under real-world conditions. ANSI/AAMI ES 60601-1 aligns with other regulatory standards, such as IEC 62133 and UL standards, to provide a comprehensive framework for battery certification.
2.5 FDA, UN/DOT, and CTIA Compliance
You must address additional regulatory standards for battery certification in the United States. The FDA categorizes medical devices by risk and sets safety standards for medical device batteries. UN 38.3 confirms lithium battery safety for transport and use. You must complete eight tests to ensure battery safety for shipping and handling. You must maintain documentation of all tests, certifications, and safety features to meet battery certification requirements.
Compliance Requirement | Description |
|---|---|
FDA | Categorizes medical devices by risk and sets US safety standards. |
UN 38.3 | Confirms lithium battery safety for transport and use. |
Testing | Eight tests required to ensure safety for shipping and handling. |
Documentation | Essential to maintain records of tests, certifications, and safety features. |
You must also consider CTIA certification for wireless medical device batteries. CTIA focuses on battery management systems and wireless communication safety. You must align your battery certification with all relevant regulatory standards to ensure patient safety and global market access.
You must integrate these regulatory standards into your battery certification process. This approach ensures your medical device battery meets all battery requirements for patient safety, quality management system effectiveness, and global market success.
Part3: Battery Certification Process Steps

3.1 Documentation and Initial Assessment
You begin the battery certification journey by preparing comprehensive documentation and conducting an initial assessment. This step sets the foundation for meeting battery requirements and ensures your medical device battery aligns with regulatory expectations. You must gather technical files, risk assessments, and design calculations. Selecting pre-certified components can minimize your testing scope and streamline the process.
Documentation Requirement | Description |
|---|---|
Pre-certified component selection | Minimizes testing scope |
Thermal management safety margins | Ensures safety in design calculations |
Documentation alignment | Must align with certification standard requirements |
Project planning | Should account for certification timelines and costs |
You also need to implement risk controls for manufacturing, documented according to ISO 14971. The level of detail varies by device class and risk level.
Tip: Align your documentation with the specific battery requirements of each certification standard to avoid costly delays.
Common pitfalls at this stage include misinterpreting IEC standards, providing incomplete risk assessments, and treating transportation and operational safety as separate issues. These mistakes can delay your project or even cause compliance failures.
Misinterpreting or selectively applying IEC standards can lead to compliance failures.
Inadequate risk assessment and safety documentation often result in delays.
Treating transportation and operational safety as separate issues complicates compliance.
3.2 Accredited Laboratory Testing
After your documentation passes the initial review, you move to accredited laboratory testing. You must select a globally recognized laboratory that meets the battery requirements for your target markets. Leading labs such as Kiwa Primara, ASCA-accredited laboratories, Eurofins Medical Device Testing, and TÜV SÜD offer comprehensive testing protocols for lithium battery packs.
Laboratory Name | Accreditation Standards | Testing Protocols |
|---|---|---|
Kiwa Primara | IEC, EN, ISO, UN, UL, MIL/RTCA | Electrical, mechanical, environmental, abuse, and safety testing (UN38.3, IEC 62133, etc.) |
ASCA-Accredited Laboratories | FDA-recognized standards | Testing methods as per ASCA requirements |
Eurofins Medical Device Testing | IEC 60601-1 | EMC testing and certification for medical devices |
TÜV SÜD | MDR requirements | Testing and certification for international market regulations |
Kiwa Primara conducts tests according to international standards such as IEC, EN, ISO, UN, UL, and MIL/RTCA. ASCA-accredited laboratories must demonstrate accreditation by recognized bodies to FDA standards. Eurofins Medical Device Testing provides EMC testing and certification for compliance with medical device regulations.
Testing protocols include electrical, mechanical, and environmental abuse tests. You must ensure your battery management systems meet all safety and performance criteria.
The cost and duration of laboratory testing vary by certification type. The table below summarizes industry benchmarks:
Certification | Avg Cost | Timeframe |
|---|---|---|
UL 1642 | $15k-$20k | 10-12w |
IEC 62133 | $6k-$10k | 6-8w |
UN38.3 | $5k-$7k | 4-6w |
KC Certification | $3,000–$5,000 | 4-8w |
CB Scheme | $4,000–$6,000 | 6-10w |
PSE Certification | $2,000–$4,000 | 4-6w |
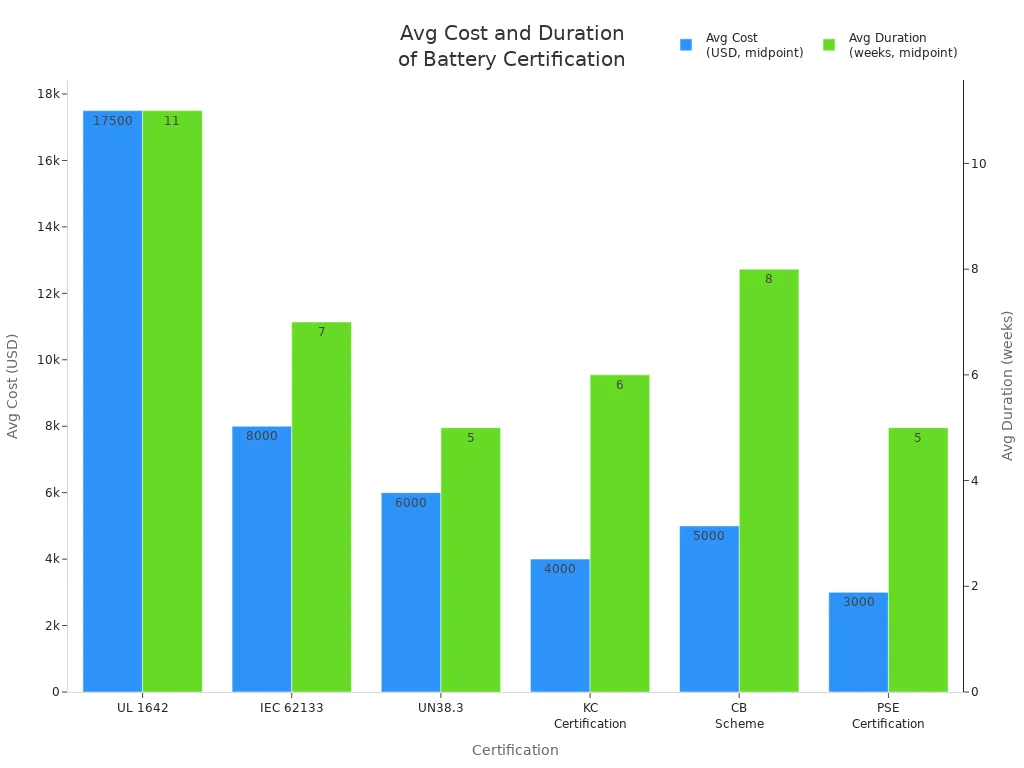
You should plan your project timeline and budget based on these benchmarks to avoid unexpected delays.
3.3 Application Submission and Review
Once you complete laboratory testing, you proceed to the application submission and review phase. This process involves several key steps:
Application Submission: You submit your battery for certification, including all required documentation and test results.
Testing Phase: The certification body reviews your submission and may conduct additional tests to verify battery requirements.
Results Analysis: Experts analyze the results to determine if your battery meets all safety and performance criteria.
Compliance Certification: If your battery passes, you receive a certificate of compliance, confirming your product meets battery pack certifications for your target markets.
You must ensure all documentation is accurate and complete to prevent delays during this phase.
3.4 Feedback and Remediation
During the review, you may receive feedback from the certification body. This feedback often highlights gaps in documentation, test results, or battery requirements. You must address these issues promptly to keep your project on track.
Review all feedback carefully and clarify any uncertainties with the certification body.
Update your documentation and testing protocols as needed.
Implement corrective actions to resolve any identified deficiencies.
Note: Proactive communication with the certification body can speed up the remediation process and reduce the risk of repeated submissions.
3.5 Sustaining Compliance
Achieving battery certification is not the end of your journey. You must sustain compliance throughout the product lifecycle. This involves regular maintenance, ongoing documentation, and periodic reviews to ensure your medical device battery continues to meet evolving battery requirements.
Best practices for sustaining compliance include:
Adhering to established standards for safety and reliability.
Conducting regular maintenance of battery systems.
Ensuring thorough documentation of compliance processes.
Following ISO/IEC 17020 and ISO/IEC 17065 standards for impartiality, competence, transparency, and traceability.
You should also perform periodic safety and compliance checks, maintain traceability, and update your processes as regulatory standards evolve. This approach helps you maintain your certifications and supports long-term market access.
Callout: Sustaining compliance protects your business from recalls, legal penalties, and market bans. It also builds trust with healthcare partners and end users.
By following these steps, you ensure your lithium battery packs meet all battery requirements for global medical device applications.
Part4: Selecting Certifications for Global Markets
4.1 Device Type and Market Mapping
You must start by mapping your medical device battery to the correct certification based on device type and target market. Each device category—such as implantable monitors, portable imaging, or surgical robotics—faces unique safety and reliability demands. For example, implantable devices require stricter standards than external monitors. The table below highlights how different standards impact safety and reliability:
Standard | Impact on Safety and Reliability |
|---|---|
ANSI/AAMI ES 60601-1 | Ensures electrical safety for devices in the US and Europe |
IEC 60086-4 | Focuses on leak resistance and stability, reducing malfunctions |
UL 2054 | Contributes to a 40% decrease in safety incidents over five years |
You must also consider the regulatory landscape in each region. The European Union requires CE marking and compliance with EMC and Low Voltage directives. The US mandates FCC and NRTL approvals. Canada, Australia, and the UK each have their own frameworks. Launching connected products globally requires careful sequencing of certifications and coordination of submissions.
Tip: Regulatory certifications validate compliance with safety, performance, and reliability standards. Selecting certified batteries enhances compatibility with global battery pack certifications.
4.2 Aligning Certification with Regulatory Standards
You need to align your battery pack certifications with the regulatory standards of each target market. Begin by understanding the legal framework in every region where you plan to launch. Implement a quality management system that meets ISO standards. Conduct thorough internal testing and work with third-party laboratories to verify compliance. Maintain strong documentation, including technical files and test results. Monitor regulatory changes and harmonize your approach to meet evolving international standards.
Understand specific market regulations.
Implement ISO-aligned quality management systems.
Engage third-party labs for compliance verification.
Maintain robust documentation.
Monitor and adapt to regulatory changes.
4.3 Integrating Certification in Product Development
Integrate certification planning early in your product development process. Early integration helps you avoid costly redesigns and certification delays. For example, a project that switched from custom-shaped cells to standard cell formats reduced lead time by 10 weeks. By considering safety and regulatory requirements during the design phase, you speed up production and reduce time-to-market.
Plan certification requirements at the concept stage.
Choose standard battery formats when possible.
Design for compliance to minimize future changes.
Note: Integrating certification early ensures your medical device battery meets global battery pack certifications and supports a smooth market launch.
Part5: Challenges and Best Practices in Battery Certification
5.1 Technical and Regulatory Hurdles
You face several technical and regulatory hurdles when certifying a medical device battery. You must ensure that batteries are removable and replaceable by end users without special tools by February 2027. Some devices qualify for exceptions, but you need to document these through a technical assessment. You also manage obligations related to conformity assessment, technical documentation, and due diligence management systems. These requirements demand a strong focus on battery traceability and iso 13485 certification throughout your process.
You must design for user-replaceable batteries.
You need to document exceptions for specific devices.
You must maintain conformity and due diligence systems.
5.2 Documentation and Traceability
You cannot achieve regulatory compliance without robust documentation and battery traceability. You need to organize your records to support audits and investigations. The following table outlines the essential documentation types you should maintain:
Documentation Type | Description |
|---|---|
Safety Documentation | Technical specifications, testing reports, quality management certificates, and compliance records. |
Technical Documentation | System specifications, circuit diagrams, thermal management plans, and safety analysis reports. |
Quality Management Documentation | Consistent manufacturing processes, documented procedures, and iso 13485 certification. |
Traceability Records | Tracks components from receipt through assembly, ensuring rapid identification of affected units. |
Application-Specific Documentation | Focuses on unique safety considerations and regulatory frameworks for each application. |
You must prioritize battery traceability and iso 13485 certification to ensure you can quickly respond to safety issues.
5.3 Ongoing Compliance Management
You need a proactive approach to ongoing compliance management for your medical device battery. You should implement routine check protocols, train staff for emergency maintenance, and provide ongoing education programs. You must maintain detailed documentation and reporting to support battery traceability. Regular skill assessments and continuous monitoring help you meet iso 13485 certification requirements.
Train staff for routine checks and emergency actions.
Maintain detailed usage and issue records for battery traceability.
Assess staff skills and update training as technology evolves.
Monitor compliance metrics to support iso 13485 certification.
Tip: Ongoing compliance management reduces risk and supports long-term market access.
5.4 Leveraging Expert Resources
You can strengthen your battery traceability and iso 13485 certification efforts by leveraging expert resources. Many manufacturers use third-party consultants for battery advisory services, reliability investigations, and regulatory guidance. The table below highlights common consulting services:
Consulting Service | Description |
|---|---|
Battery Advisory Services | FMEA, reliability, safety investigations, and manufacturing plant audits. |
Independent power source evaluations and client confidentiality. | |
US FDA Consulting | Device classification, registration, premarket clearance, and compliance support. |
Emergo by UL |
You should engage these experts to enhance battery traceability and maintain iso 13485 certification for your medical device battery.
You can achieve global market success by following a structured approach to battery certification. Start with strong documentation, accredited testing, and ongoing compliance management. Adhering to regulatory standards protects your business and builds trust with partners. For continued regulatory compliance, take these steps:
Develop sustainability reports to show your commitment to battery life cycle goals.
Build compliance frameworks for due diligence and material tracking.
Partner with suppliers early to validate and collect essential data.
Staying proactive ensures your medical device battery remains safe, reliable, and market-ready.
FAQ
What lithium battery chemistries are most common in medical devices?
You often use LiFePO4 Lithium battery, NMC Lithium battery, LCO Lithium battery, and LMO Lithium battery in medical devices. Each chemistry offers different platform voltage, energy density, and cycle life. The table below compares these properties:
Chemistry | Platform Voltage (V) | Energy Density (Wh/kg) | Cycle Life (cycles) |
|---|---|---|---|
LiFePO4 Lithium | 3.2 | 90–120 | 2000+ |
NMC Lithium | 3.7 | 150–220 | 1000–2000 |
LCO Lithium | 3.7 | 150–200 | 500–1000 |
LMO Lithium | 3.7 | 100–150 | 500–1000 |
How do you ensure regulatory compliance for lithium battery packs?
You must follow regulatory standards such as IEC 62133, UL 2054, and ISO 13485. You need to document all processes, use accredited labs for testing, and maintain traceability from component sourcing to final assembly.
What is the typical certification timeline for a medical device battery?
You should expect the battery certification process to take 4 to 12 weeks. The timeline depends on the chemistry, device type, and the number of required tests. Early planning helps you avoid delays.
Why is traceability important in lithium battery pack manufacturing?
Traceability lets you track every component and process step. This practice supports fast recalls, quality audits, and regulatory compliance. You reduce risk and improve safety for end users.
Can you use the same battery certification for different global markets?
You often need separate certifications for each region. For example, CE marking is required in Europe, while UL standards apply in North America. Align your certification strategy with your target markets.




